September 22, 2023 Release Highlights Copied
Practice Insights is pleased to announce updates in the application to support the Merit-Based Incentive Payment System (MIPS).
National Comprehensive Cancer Network (NCCN) Distress Thermometer Dashboard
The new NCCN Distress Thermometer dashboard is a tracking mechanism that empowers your practice with invaluable insights. This dashboard is integrated with iKnowMed Generation 2, offering a comprehensive overview of patients’ distress levels and ensuring every documented distress level is captured and visualized on a weekly basis.
Use the NCCN Distress Thermometer Dashboard to meet the requirements of MIPS Measure #487: Screening for Social Drivers of Health (planned for release in early 2024), which will assess the total number of patients who have completed a screening for health-related social needs.
NCCN Distress Thermometer Dashboard Features
To use the NCCN Distress Thermometer dashboard:
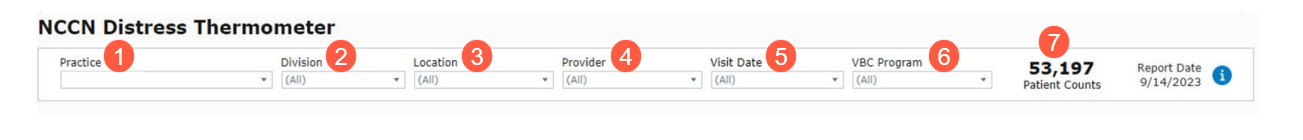
- Make one or more selections (callouts 1-6) to filter the dashboard by:
- Practice
- Division
- Location
- Provider
- Visit Date
- VBC Program
- The dashboard includes a dynamic Patient Count (callout 7) that adjusts based on your filters.
- A patient is only counted once per location within the dashboard with their most recent screening displayed. This means if a patient is treated at multiple locations in the same week, they will be displayed for each location.
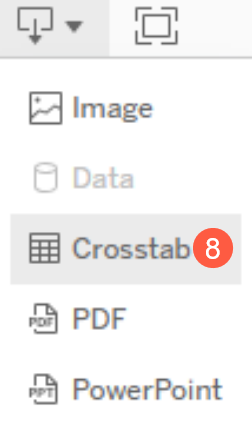
- To export the report, click the Export Dropdown Functionality button and select the Crosstab option (callout 8).
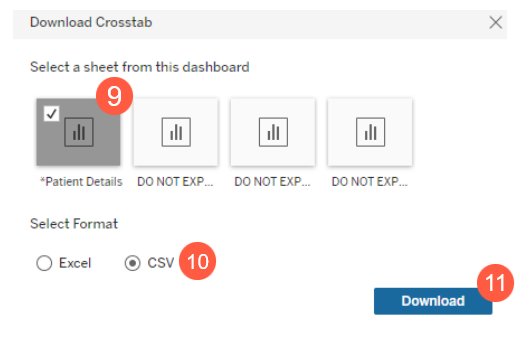
- From the Download Crosstab window, select Patient Details (callout 9), choose your preferred document format (callout 10), and click the Download button (callout 11) to complete the data export.
NOTE: Choose the CSV format for the most optimal experience. Once downloaded, you can save the file in Excel format.
MIPS Quality Measures Dashboard
The dashboard has also been updated to reflect the below refinement(s) for:
| Measure | Description | |
|---|---|---|
| MIPS #453: Percentage of Patients who Died from Cancer Receiving Systemic Cancer-Directed Therapy in the Last 14 Days of Life (Inverse Measure) |
|
Practice-level results will be minor; performance rates for individual clinicians will be more significant due to the attribution update to associate numerator criteria with a single prescribing clinician. |
Status of Dashboard Updates for 2023
CMS releases updates to the quality measure specifications annually for the MIPS program and bi-annually for the OCM program. These measure specifications have been reviewed, and Practice Insights is working on the required technical updates to align with the revised requirements. McKesson will continue to notify practices once measures have been updated based on the current 2023 requirements. The measures listed below either have no 2023 update required or have been updated.
| Measure | Description |
|---|---|
MIPS Quality Measures Dashboard |
|
| #047 | Advance Care Plan |
| #104 | Adjuvant Hormonal Therapy for High-Risk or Very High-Risk Prostate Cancer |
| #130 | Current Medications Documentation |
| #134 | Preventive Care Screening: Screening for Depression and Follow-Up Plan |
| #143 | Pain Intensity Quantified |
| #144 | Pain Care Plan |
| #226 | Tobacco Screening and Cessation |
| #236 | Controlling High Blood Pressure |
| #374 | Closing the Referral Loop |
| #451 | RAS (KRAS and NRAS) Gene Mutation Testing Performed for Patients with Metastatic Colorectal Cancer who Receive Anti-epidermal Growth Factor Receptor (EGFR) Monoclonal Antibody Therapy |
| #452 | Percentage of Adult Patients (aged 18 or older) with Metastatic Colorectal Cancer and RAS (KRAS & NRAS) Gene Mutation Spared Treatment with Anti-EGFR Monoclonal Antibodies |
| #453 | Percentage of Patients who Died from Cancer Receiving Systemic Cancer-Directed Therapy in the Last 147 Days of Life (Inverse Measure) |
| #457 | Patients Admitted to Hospice for < 3 Days |
| PIMSH#1 | Advance Care Planning in Stage 4 Disease |
| PIMSH#2 | Utilization of GCSF in Metastatic Colon/Rectal Cancer (Inverse Measure) |
| PIMSH#4 | Patient-Reported Pain Improvement |
| PIMSH#8 | Mutation Testing for Lung Cancer |
| PIMSH#9 | Supportive Care Drug Utilization in the Last 14 Days of Life (Inverse Measure) |
| PIMSH#10 | Hepatitis B Serology Testing for Prophylactic Treatment Prior to Receiving Anti CD20 Targeting Drugs |
| PIMSH#15 | Antiemetic Therapy for Low-and-Minimal-Emetic Risk Antineoplastic Agents in the Infusion Center-Avoidance of Overuse (Inverse Measure) |
| PIMSH#16 | Appropriate Antiemetic Therapy for High-and Moderate-Emetic-Risk Antineoplastic Agents in the Infusion Center |
