November 15, 2024 Release Highlights Copied
Practice Insights is pleased to announce updates in the application to support the Enhancing Oncology Model (EOM).
EOM Performance Dashboard
The pie charts for the different EOM measures now feature an improved, user-friendly data label display.
- Before: Static data labels made it difficult to read and match the data values to the correct pieces of the pie chart.
- Post-Release: Each pie slice will have an interactive hover effect so users can hover over any segment to see a clear view of the associated data label and value.
This enhancement provides an improved user experience, allowing users to quickly and accurately identify the information for each measure.
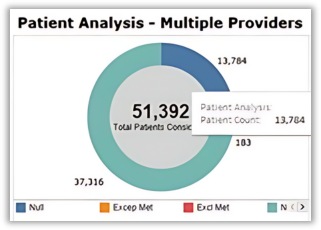
Status of Dashboard Updates for 2024
CMS releases updates to the quality measure specifications annually for the MIPS program. These measure specifications have been reviewed, and Practice Insights is working on the required technical updates to align with the revised requirements. McKesson will continue to notify practices once measures have been updated based on the current 2024 requirements. The measures listed below either have no 2024 update required or have been updated.
| Measure | Description |
|---|---|
MIPS Quality Measures Dashboard |
|
| #001 | Diabetes Hemoglobin A1c (HbA1c) Poor Control (>9%) (Inverse Measure) |
| #047 | Advance Care Plan |
| #104 | Adjuvant Hormonal Therapy for High Risk or Very High Risk Prostate Cancer |
| #128 | BMI (This measure is no longer a MIPS Traditional Measure) |
| #130 | Current Medications |
| #134 eCQM | Preventive Care Screening: Screening for Depression and Follow-Up Plan |
| #134 CQM / Registry | Preventive Care Screening: Screening for Depression and Follow-Up Plan |
| #143 | Pain Intensity Quantified |
| #144 | Pain Care Plan |
| #226 | Preventive Care and Screening: Tobacco Use: Screening and Cessation Intervention |
| #236 | Controlling High Blood Pressure |
| #238 | Use of High-Risk Medications in Older Adults |
| #374 | Closing the Referral Loop |
| #450 | Trastuzumab Received by Patients with AJCC Stage I (T1c), II and III and HER2+ Breast Cancer Receiving Adjuvant Chemotherapy |
| #451 | RAS (KRAS & NRAS) Testing Performed for Patients with Metastatic Colorectal Cancer who Received Anti-EGFT Monoclonal Antibody Therapy |
| #452 | Percentage of Adult Patients (aged 18 or older) with Metastatic Colorectal Cancer and RAS (KRAS & NRAS) Gene Mutation Spared Treatment with Anti-EGFR Monoclonal Antibodies |
| #453 | Percentage of Patients Who Died from Cancer Receiving Systemic Cancer-Directed Therapy in the Last 14 Days of Life (Inverse Measure) |
| #457 | Percentage of Patients Who Died from Cancer Admitted to Hospice for Less than 3 days (Inverse Measure) |
| #462 | Bone Density Evaluation for Patients with Prostate Cancer and Receiving Androgen Deprivation Therapy |
| #487 | Screenings for Social Drivers of Health |
| PIMSH #1 | Advance Care Planning in Stage 4 Disease |
| PIMSH #2 | Utilization of GCSF in Metastatic Colon/Rectal Cancer (Inverse Measure) |
| PIMSH #4 | Patient-Reported Pain Improvement |
| PIMSH #9 | Supportive Care Drug Utilization in the Last 14 Days of Life (Inverse Measure) |
| PIMSH #10 | Hepatitis B Serology Testing for Prophylactic Treatment Prior to Receiving Anti-CD20 Targeting Drugs |
| PIMSH #13 | Proportion of Stage IV nsNSCLC Patients Tested for Actionable Biomarkers and Received Mutation-Targeted Therapy (EGFR, BRAF, ROS1, ALK, PD-L1) |
| PIMSH #15 | Antiemetic Therapy for Low-and-Minimal-Emetic Risk Antineoplastic Agents in the Infusion Center-Avoidance of Overuse (Inverse Measure) |
| PIMSH #16 | Appropriate Antiemetic Therapy for High- and Moderate-Emetic-Risk Antineoplastic Agents in the Infusion Center |
