Adverse Events: Expanded attribution options Copied
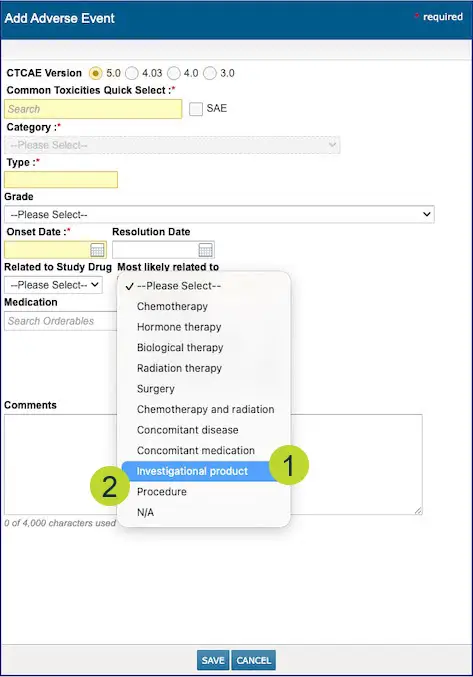
Two new options are available in the “Most Likely Related to” dropdown when documenting Adverse Events (callouts 1 and 2):
- Investigational Product
- Procedure
What’s changing
Previously, these attribution options were not available when documenting why a patient experienced an adverse event.
You can now select and save either option to the patient’s chart under Clinical Profile > Observation > Adverse Events.
Why this matters
These additions provide more precise attribution for adverse events, particularly for research protocols and procedure-related complications.
